Advertisements
Advertisements
प्रश्न
उत्तर
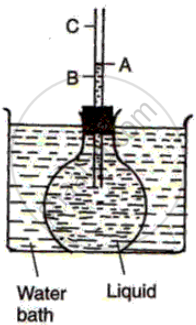
Fill a round bottom flask with a coloured liquid up to the brim. Fit it with a one holed cork and pass a narrow glass tube through it. Mark the level of the liquid in the glass tube as A. Place the flask in a water bath and heat the bath. You will see that the level of liquid of water starts falling to level Band then it rises to level C. In this, AC is the apparent expansion and is due to expansion of the flask due to heating which provides more space for the liquid. Hence the level of liquid drops. As soon as the liquid also starts getting heated up, it expands and the level of liquid rises. BC is the real expansion. So the actual expansion of the water is the sum of AC and AB.
APPEARS IN
संबंधित प्रश्न
What is matter? What is it composed of?
What do you understand by thermal expansion of a substance?
State one application of thermal expansion of liquids.
Describe an experiment to showthat air expands on heating.
Describe an experiment to show that same volume of different liquids heated to same rise in temperature expand by different amounts.
100 ml of each of the following liquid is heated from 10°C to 50°C. Which will expand more : (a) water (b) benzene (c) alcohol?
An iron washer is heated. State the effect on its mass.
The diagram shows a sketch graph showing a change in volume of a given mass of ice at -5°C to water at 15°C.
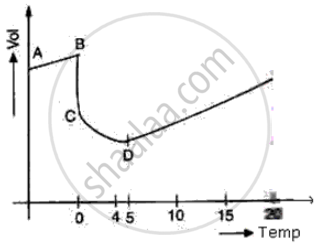
(i) Why does the temperature along BC remain constant?
(ii) When is the volume of water minimum?
(iii) At what temperature does water have maximum density? Explain.
What difference do you think heating the solid will make in their molecules?
