Advertisements
Advertisements
प्रश्न
Three organic compounds A, B and C have the following molecular formulae: C4H8O2
Which compound contains an alcohol group? Write its name and structural formula.
उत्तर
The organic compound 'B' comprises an alcohol group. Butanol is the name of the compound, and its formula is C4H9OH.
Its structural formula is as follows:
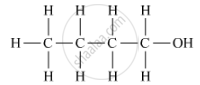
APPEARS IN
संबंधित प्रश्न
Give two properties of ethanol.
The compound formed when ethene reacts with Hydrogen is ________. (CH4, C2H6, C3H8)
State one relevant observation for the following
When the gaseous product obtained by dehydration of ethyl alcohol is passed through bromine water.
Name the addition product formed between ethene and water.
An organic compound 'P' is a constituent of wine. 'P' on reacting with acidified K2Cr2O7 forms another compound 'Q'. When a piece of sodium is added to 'Q' a gas 'R' evolves which burns with a pop sound. Identify P, Q and R and write the chemical equations of the reactions involved.
In a tabular form, differentiate between ethanol and ethanoic acid under the following heads:
(i) Physical state
(ii) Taste
(iii) NaHCO3 test
(iv) Ester test
Write a chemical reaction to show the dehydration of ethanol.
Write a balanced chemical equation for the following:
Ethanol reacts with sodium at room temperature
Name an organic compound which is dehydrated to produce ethene.
Give the balanced chemical equation of the following reaction:
Combustion of ethanol.
