Advertisements
Advertisements
प्रश्न
Water is used as a cooling agent.
उत्तर
Water has high specific heat. Water neither heats up nor cools down quickly. This property makes water an excellent cooling agent.
APPEARS IN
संबंधित प्रश्न
Give reason of Water pipes burst in severe winter.
Give reason of Ice floats on water.
Consider the situation of the previous problem. Assume that the temperature of the water at the bottom of the lake remains constant at 4°C as the ice forms on the surface (the heat required to maintain the temperature of the bottom layer may come from the bed of the lake). The depth of the lake is 1.0 m. Show that the thickness of the ice formed attains a steady state maximum value. Find this value. The thermal conductivity of water = 0.50 W m−1°C−1. Take other relevant data from the previous problem.
A calorimeter contains 50 g of water at 50°C. The temperature falls to 45°C in 10 minutes. When the calorimeter contains 100 g of water at 50°C, it takes 18 minutes for the temperature to become 45°C. Find the water equivalent of the calorimeter.
How do fishes survive in frozen lakes?
A deep pond of water has its top layer frozen. What will be the likely temperature of water layer at the bottom of the pond?
What are hot spots? How can you extract energy from a hot spot, if it does not come in contact with underground water?
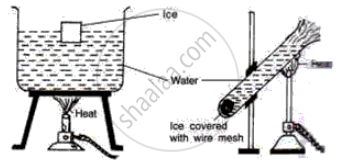
Study the following diagrams and write down your observations.