Advertisements
Advertisements
प्रश्न
What happens to the average kinetic energy of the molecules as ice melts at 0°C?
उत्तर
Average Kinetic Energy of molecules increases.
APPEARS IN
संबंधित प्रश्न
Name the law on which principle of Calorimetry is based
Explain the melting point ?
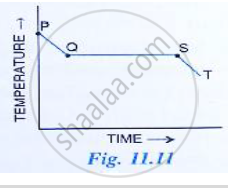
Fig 11. 11 shows the variation in temperature with time when some wax cools from the liquid phase to the solid phase.
(i) In which part of the curve, the wax is in liquid phase?
(ii) What does the part QS of the curve represent?
(iii) In which part of the curve, the wax will be the in the liquid as well as solid phase?
(iv) In which part of the curve, the wax is in solid phase?
A mass of 200 g of mercury at 100°C is poured into water at 20°C. If the final temperature of the mixture is 25°C, find the mass of water. (Sp. heat cap. of mercury is 140 J/kgK).
Give scientific reasons for the following:
Sand mixed with salt is often spread over the icy roads in winter.
40 g of ice at -16°C is dropped into water at 0°C, when 4 g of water freezes into ice. If specific heat capacity of ice is 2100 J/kg°C, what will be the latent heat of fusion of ice?
Which contains more heat: 1 G water at 100°C or 1 g steam at 100°C? Give reason.
Why does atmospheric temperature fall after hail storm?
Explain briefly, how you would determine the specific heat capacity of a liquid?
Calculate the heat energy that will be released when 5.0 kg of steam at 100°C condenses to form water at 100°C. Express your answer in S.I. unit. (Specific latent heat of vaporization of steam is 2268 kj/kg.)
