Advertisements
Advertisements
प्रश्न
What happens when bleaching powder reacts with dilute sulphuric acid? Give equation of the reaction involved.
उत्तर
When bleaching powder reacts with dilute sulphuric acid, all the chlorine present in it is liberated. This chlorine acts as a bleaching agent.
CaOCl2 + H2SO4 → CaSO4 + Cl2 + H2O
APPEARS IN
संबंधित प्रश्न
Give the molecular formula of bleaching powder.
Name one compound of calcium which is used for removing the colour of a coloured cloth.
What is "baking powder"? How does it make the cake soft and spongy?
State two uses each of the following compounds:
Chlorine
What is bleaching powder?
Consider the following substance:
NaCl, Ca(OH)2, NaHCO3, NH3, Na2CO3, H2O, Cl2, CO2, CaSO4.2H2O, 2CaSO4.H2O, CaOCl2
Which two substance combine to form bleaching powder?
Brine is an ____________.
CaOCl2 will liberate Cl2 gas in the presence of
(i) CO2
(ii) HCl
(iii) CO
(iv) NO
The reaction between MnO2 with HCl is depicted in the following diagram. It was observed that gas with bleaching abilities was released.
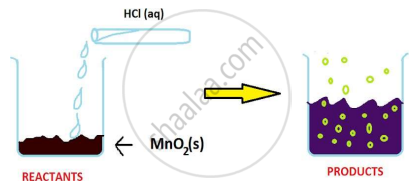
The chemical reaction between MnO2 and HCl is an example of:
Chlorine gas reacts with ____________ to form bleaching powder.
