Advertisements
Advertisements
प्रश्न
What is the role of concentrated Sulphuric acid when it is heated with Ethanol at 443 K. Give the reaction involved.
उत्तर
Sulphuric acid acts as dehydrating agent
\[\ce{C2H5OH ->[Conc H2SO4,443K] C2H4 + H2O}\]
APPEARS IN
संबंधित प्रश्न
Give other two names of ethanol.
Complete the following equation:
`CH_3 CH_2 OH` 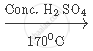
Give the lab. Prepared of ethyl alcohol .
With a labelled diagram describe an activity to show the formation of an ester.
How does ethanol reacts with
(a) Metallic sodium
(b) Acetic acid
(c) Conc.H2S04
What are hydrocarbons? Give one example.
Generally _______ is called spirit.
Colorless ethanol is in liquid state at room temperature.
An organic compound ‘A’ is widely used as a preservative and has the molecular formula C2H4O2. This compound reacts with ethanol to form a sweet-smelling compound ‘B’.
Write the chemical equation for its reaction with ethanol to form compound ‘B’.
Give the balanced chemical equation of the following reaction:
Combustion of ethanol.
