Advertisements
Advertisements
प्रश्न
Why are Zn2+ ions colourless while Ni2+ ions are green in colour?
उत्तर
Zn2+ ions are colourless due to the absence of any unpaired electrons in its 3d-orbital.
Whereas, Ni2+ ions are green in colour due to the presence of 2 unpaired electrons d-d transitions are possible.
Zn2+ = 3d10 4s0
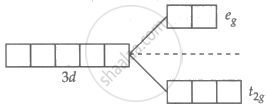
No unpaired electron in Zn2+.
∴ No d-d transition, colourless.
Ni2+ = 3d8 4s0
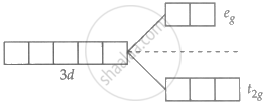
Unpaired electron present in Ni2+.
∴ d-d transition possible and are coloured.
APPEARS IN
संबंधित प्रश्न
Fill in the blanks by choosing the appropriate word(s) from those given in the brackets:
(two, four, sec-1, diamagnetic, acetaldehyde, mol-1L sec-1, paramagnetic, formaldehyde, acetone, ethanol)
The transition metals show ______ character because of the presence of unpaired electrons while Cu+ is ______ because its electronic configuration is [Ar]3d10.
How would you account for the following:
Metal-metal bonding is more extensive in the 4d and 5d series of transition elements than the 3d series.
