Advertisements
Advertisements
प्रश्न
Would you cool a bucket of water more quickly by placing it on ice or by placing ice in it ? Give reasons for your answer.
उत्तर
We can cool a bucket of water quicker when we keep ice inside the bucket because ice cubes absorb more and more heat from the liquid system in which it is present, cooling the liquid system quickly. But when we put water in ice, the ice comes in contact with the bucket and absorbs the latent heat from both the water and bucket. Therefore, the effectiveness of cooling is reduced.
APPEARS IN
संबंधित प्रश्न
Why does our palm feel cold when we put some acetone or petrol or perfume on it?
Explain why in hot summer days water remains cool in earthen pots.
Define the following term Freezing.
Why are we able to sip hot tea or milk faster from a saucer rather than from a cup ?
How will you demonstrate that water vapour is present in air ?
What is evaporation ? State the various factors which affect evaporation.
Give a geographical reason:
Air becomes saturated.
Can you suggest a method to collect water from sea water?
Look at Fig. 1.3 and suggest in which of the vessels A,B, C or D the rate of evaporation will be the highest? Explain.
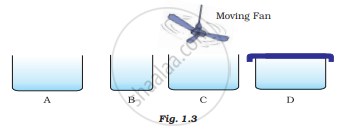
Evaporation is a fast process and occurs only at the surface of the liquid.
