Advertisements
Advertisements
प्रश्न
Write balanced chemical equations to show how SO3 is converted to Sulphuric acid in the contact process.
उत्तर
SO3 + H2SO4 → H2S2O7 oleum or pyrosulphuric acid
H2S2O7 + H2O → 2H2SO4
APPEARS IN
संबंधित प्रश्न
Quicklime is not used to dry \[\ce{HCl}\] gas because _____.
Describe the reaction that show
Concentrated sulphuric acid is a non-volatile acid.
Sulphuric acid can be used to prepare a number of gases in the laboratory. Write balanced equation for the reaction from which the folllowing gas are obtained, using dilute sulphuric acid as one of the reactant : Carbon dioxide
What would you see, when a crystal of CuSO4.5H2O is placed in concentrated sulphuric acid? Give reason to explain your observation.
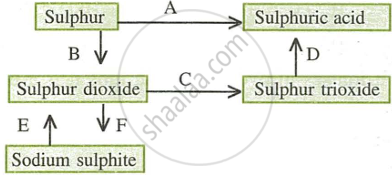
- Name the catalyst which helps in the conversion of sulphur dioxide to sulphur trioxide in step C.
- In the contact process for the manufacture of sulphuric acid, sulphur trioxide is not converted to sulphuric acid by reacting it with water. Instead a two-step procedure is used. Write the equations for the two steps involved in D.
- What type of substance will liberate sulphur dioxide from sodium sulphite in step E?
- Write the equation for the reaction by which sulphur dioxide is converted to sodium sulphite in step F.
Which property of sulphuric acid accounts for its use as a dehydrating agent ?
Give balanced chemical equation to prepare the following salt:
Sodium sulphate using dilute sulphuric acid
The gas evolved when dilute sulphuric acid reacts with iron sulphide.
The salt is formed when concentrated sulphuric acid reacts with KNO3 above 200°C.
Hydrogen chloride gas is prepared in the laboratory by the action of concentrated sulphuric acid on sodium chloride.
State the method of collection of the gas formed above.
