Advertisements
Advertisements
प्रश्न
Write the cell reaction and calculate the e.m.f of the following cell at 298 K:
`Sn(s) | Sn^(2+) (0.004 M) || H^(+) (0.020 M) | H_2 (g) ("1 bar") | Pt(s)`
(Given: `E_(Sn^(2+)"/"Sn)^0 = -0.14` V)
उत्तर
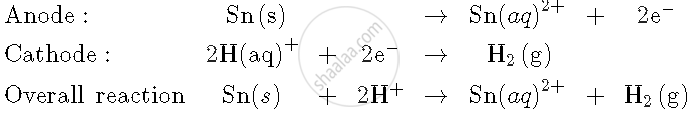
`E_"cell"^0 = E_(Sn"/"Sn^(+2))- 0.0`
= -(-0.14)
= 0.14 V
`E_"cell = E_(cell)^0 - 0.059/2 log ([H_2][Sn^(+2)])/[H^(+)]^2`
`= 0.14 - 0.059/2 log ((1)(0.004))/(0.02)^2`
`= 0.14 - 0.059/2 log (10)`
= 0.14 - 0.0295
= +0.1105 V
APPEARS IN
संबंधित प्रश्न
How would you determine the standard electrode potential of the system \[\ce{Mg^{2+} | Mg}\]?
Consult the table of standard electrode potentials and suggest three substances that can oxidise ferrous ions under suitable conditions.
Calculate the standard cell potential of a galvanic cell in which the following reaction takes place:
\[\ce{2Cr_{(s)} + 3Cd^2+_{( aq)}-> 2Cr^{3+}_{( aq)} + 3Cd}\]
Calculate the ΔrGΘ and equilibrium constant of the reaction.
Account for the following:
E° value for the Mn3+/Mn2+ couple is highly positive (+1.57 V) as compare to Cr3+/Cr2+.
In a hydrogen-oxygen fuel cell, combustion of hydrogen occurs to:-
In a galvanic cell, current flows ______
What is the standard free energy change for the following reaction at room temperature? Is the reaction spontaneous?
\[\ce{Sn(s) + 2Cu^{2+}(aq) -> Sn^{2+}(aq) + 2Cu+(s)}\]
Galvanization is applying a coating of ______.
Calculate the standard cell potential of a galvanic cell in which the following reaction takes place:
\[\ce{Fe^2+_{( aq)} + Ag+_{( aq)} -> Fe^3+_{( aq)} + Ag_{(s)}}\]
Calculate the ΔrGθ and equilibrium constant of the reaction.
