Advertisements
Advertisements
प्रश्न
Write the equation to show the reaction between the following:
Between copper and concentrated nitric acid.
उत्तर
\[\ce{Cu + 4HNO3 ->[\Delta] Cu(NO3)2 + 2NO2 + 2H2O}\]
APPEARS IN
संबंधित प्रश्न
Name the gas that is produced in the given case:
The action of cold and dilute nitric acid on copper
What is aqua fortis?
- Write a balanced chemical equation for the laboratory preparation of nitric acid.
- In the preparation of nitric acid from \[\ce{KNO3}\], concentrated hydrochloric acid is not used in place of concentrated sulphuric acid. Explain why?
- Conc. nitric acid prepared in laboratory is yellow in colour. Why? How is this colour removed?
- Give reasons for the following:
In the laboratory preparation of nitric acid, the mixture of concentrated sulphuric acid and sodium nitrate should not be heated very strongly above 200°C.
In the preparation of nitric acid from KNO3 concentrated hydrochloric acid is not used in place of concentrated sulphuric acid. Explain why?
Conc. Nitric acid prepared in laboratory is yellow in colour why? How is this colour removed?
Write the balanced equation for the following:
Action of heat on AgNO3
Choose the correct word from the given options to complete the sentence :
Sulphur can be converted to sulphuric acid using __________ nitric acid.
Write equation to show the reaction between the following:
Copper oxide and dilute nitric acid.
Fill in the blank using the appropriate words given below:
Hot, concentrated nitric add reacts with sulphur to form ______.
The diagram given below is a representation of the Industrial preparation of Nitric acid by Ostwald’s process. With respect to the process answer the following questions:
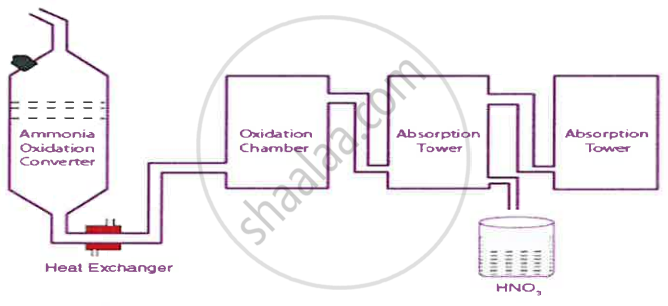
- Write the temperature and the catalyst required during the catalytic oxidation of ammonia.
- Give balanced chemical equation for the reaction occurring duringthe conversion of nitrogen dioxide to nitric acid.
