Advertisements
Advertisements
प्रश्न
Write equation to show the reaction between the following:
Copper and concentrated nitric acid.
उत्तर
\[\ce{Cu + 4HNO3 ->[\Delta] Cu(NO3)2 + 2NO2 + 2H2O}\]
APPEARS IN
संबंधित प्रश्न
Give a balanced chemical equation for Action of conc. Nitric acid on Sulphur
Name the gas that is produced in the given cases :
Sulphur is oxidized by concentrated nitric acid.
What is fixation of Nitrogen?
Nitric acid cannot be concentrated beyond 68% by the distillation of a dilute solution of \[\ce{HNO3}\]. State the reason.
Name the Following:
Product obtained by the catalytic oxidation of ammonia.
Write equation to show the reaction between the following:
Copper oxide and dilute nitric acid.
Write a balanced equation for following :
Action of conc. Nitric acid on sulphur
From the formulae listed below, Choose, one, corresponding to the salt having the given description:
AgCl, CuCO3, CuSO4.5H2O, KNO3, NaCl, NaHSO4, Pb(NO3)2, ZnCO3, ZnSO4, 7H2O.
On heating this, salt changes from green to black.
Convert the following reaction into a balanced chemical equation:
Ammonia to nitric oxide using oxygen and platinum catalyst.
The diagram given below is a representation of the Industrial preparation of Nitric acid by Ostwald’s process. With respect to the process answer the following questions:
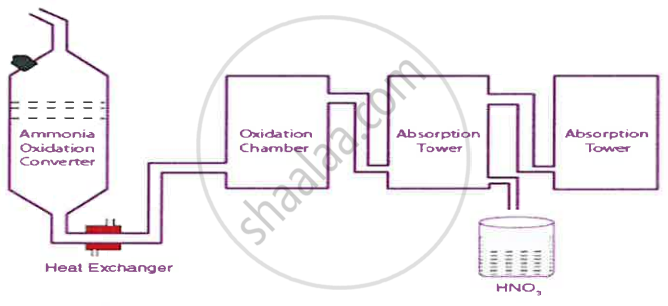
- Write the temperature and the catalyst required during the catalytic oxidation of ammonia.
- Give balanced chemical equation for the reaction occurring duringthe conversion of nitrogen dioxide to nitric acid.
