Advertisements
Advertisements
प्रश्न
Write the observation for the following:
A paper dipped in potassium permanganate solution is put on the mouth of a test tube containing sulphur dioxide gas.
उत्तर
Purple colour of potassium permanganate gets discharged.
APPEARS IN
संबंधित प्रश्न
Name oxide of one metal which is reduced by (give equation): Electrolytic reduction
Name a non-metallic element which has a metallic lustre
Name a non-metallic element which is a conductor of electricity.
Write balanced equation for the following reaction:
Reduction of iron (III) oxide by carbon monoxide.
Name the following :
A metal oxide that can be reduced by hydrogen
Answer the following question with respect by hydrogen : Identify the components of the electrolyte other than pure alumina and the role played by each.
Answer the following question with respect by hydrogen :
Explain why powdered coke is sprinkled over than electrolytic mixture.
- A to F below relate to the source and extraction of either zinc or aluminium:
- Bauxite
- Coke
- Cryolite
- Froth floatation
- Sodium hydroxide solution
- Zinc blende
- Write down the three letters each from the above list which are relevant to
- Zinc
- Aluminium
- Fill in the blanks using the most appropriate words from A to F.
- The ore from which aluminium is extracted must first be treated with ………………. so that pure aluminium oxide can be obtained.
- Pure aluminium oxide is dissolved in ….. to make a conducting solution.
iii. Write the formula of cryolite.
Give the equation for the reduction of Iron (III) oxide.
The following sketch represents the electroplating of an Iron cup with Nickel metal.
Study the diagram and answer the following questions:
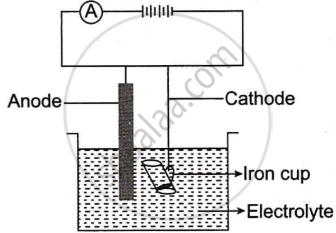
- During electroplating, the iron cup is placed at the cathode. Why?
- Name the ion that must be present in the electrolyte.
- State one condition that is necessary to ensure that the deposit is smooth, firm and even.
- Write the reaction taking place at the cathode.
- What change would you observe at the anode?
