Advertisements
Advertisements
प्रश्न
Write scientific reason.
Atomic radius goes on increasing down a group.
उत्तर
Atomic radius increases as we move from top to bottom in a group of the periodic table because a new shell of electrons is added to the atoms at every step. As the number of shells in the atoms increases gradually due to which the size of atoms also increases. As the size of the atoms increases which leads to an increase in the atomic radius of an atom.
APPEARS IN
संबंधित प्रश्न
Why is the size of neon greater than fluorine?
Why is the size of sodium is greater than magnesium?
Which is greater in size Fe2+ or Fe3+?
Fill in the blanks.
Size of the atoms ______ from left to right across a period and ______ on descending in a group of normal elements.
What happens to the atomic size of elements on moving from left to right in a period?
With reference to the variation of properties in the Periodic Table, which of the following is generally true?
Atomic size increases from left to right across a period.
The following questions refer to the Periodic Table.
What happens to the atomic size of elements moving from top to bottom of a group?
Moving from left to right, the size of the atom decreases.
The size of an atom depends on the number of valence electrons.
Write scientific reason.
In same period, boron and oxygen elements have different atomic size.
Carbon belongs to the second period and Group 14. Silicon belongs to the third period and Group 14. If the atomic number of carbon is 6, the atomic number of silicon is ______
Which of the following is the correct order of atomic size?
Which of the following gives the correct increasing order of the atomic radii of O, F, and N?
When an atom of iodine becomes an iodine ion (I–) the radius will ______
Which of the following gives the correct increasing order of the atomic radii of O, F and N?
Which of the following are the characteristics of isotopes of an element?
- Isotopes of an element have same atomic masses
- Isotopes of an element have same atomic number
- Isotopes of an element show same physical properties
- Isotopes of an element show same chemical properties
Atomic number of a few elements are given below
10, 20, 7, 14
- Identify the elements
- Identify the Group number of these elements in the Periodic Table
- Identify the Periods of these elements in the Periodic Table
- What would be the electronic configuration for each of these elements?
- Determine the valency of these elements
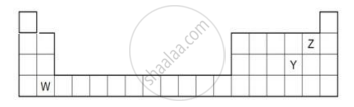
The diagram below shows part of the periodic table.
- Which elements would react together to form covalent compounds?
- Between the two elements W and Z, which will have a bigger atomic radius? Why?
Arrange the following in order of increasing radii:
Mg2+, Mg, Mg+
Explain your choice.
