Advertisements
Advertisements
प्रश्न
Write short note on metal excess and metal deficiency defect with an example.
उत्तर
Metal excess defect arises due to the presence of more metal ions as compared to anions. Alkali metal halides NaCl, KCl show this type of defect. The electrical neutrality of the crystal can be maintained by the presence of anionic vacancies equal to the excess metal ions (or) by the presence of extra cation and electrons present in the interstitial position.

Metal Excess Defect
For example, when NaCl crystals are heated in the presence of sodium vapour, Na+ ions are formed and are deposited on the surface of the crystal. Chloride ions (Cl–) diffuse to the surface from the lattice point and combines with Na+ ions.
The electron lost by the sodium vapour diffuses into the crystal lattice and occupies the vacancy created by the Cl– ions. Such anionic vacancies which are occupied by unpaired electrons are called F centers. Hence, the formula of NaCl which contains excess Na+ ions can be written as Na1+xCl.
Metal deficiency defect: Metal deficiency defect arises due to the presence of less number of cations than the anions. This defect is observed in a crystal in which, the cations have variable oxidation states. For example, in FeO crystal, some of the Fe2+ ions are missing from the crystal lattice.
To maintain electrical neutrality, twice the number of other Fe2+ ions in the crystal is oxidized to Fe3+ ions. In such cases, the overall number of Fe2+ and Fe3+ ions is less than the O2- ions. It was experimentally found that the general formula of ferrous oxide is FexO, where x ranges from 0.93 to 0.98.
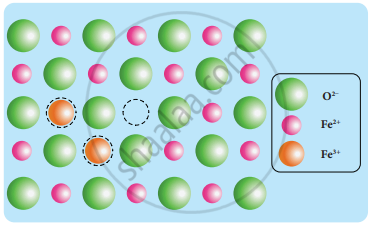
Metal Deficiency Defect
APPEARS IN
संबंधित प्रश्न
Schottky defect in a crystal is observed when
The cation leaves its normal position in the crystal and moves to some interstitial position, the defect in the crystal is known as ____________.
Assertion: due to Frenkel defect, density of the crystalline solid decreases.
Reason: in Frenkel defect cation and anion leaves the crystal.
The crystal with a metal deficiency defect is ____________.
What are point defects?
Explain Schottky defect.
Write a note on Frenkel defect.
