Advertisements
Advertisements
प्रश्न
Write the structure of all possible dipeptides which can be obtained from glycine and alanine.
Give the structure of all the possible dipeptides formed when the following two amino acids form a peptide bond.
| Alanine |
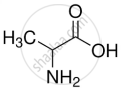 |
| Glycine |
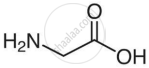 |
उत्तर
\[\begin{array}{cc}
\ce{\underset{(Glycine)}{H2N - CH2 - COOH} + H2N - CH - COOH ->[-H2O] H2N - CH2 - CONH - CH - COOH}\\
\phantom{......................}|\phantom{...........................................}|\phantom{...}\\
\phantom{.........................}\ce{\underset{(Alanine)}{CH3}}\phantom{...................................}\ce{\underset{(Glycyl alanine)}{CH3}}\phantom{}
\end{array}\]
\[\begin{array}{cc}
\ce{H2N - CH - COOH + \underset{(Glycine)}{H2N - CH2 - COOH} ->[][-H2O] H2N - CH - CONH - CH2 - COOH}\\
\phantom{..}|\phantom{..................................................}|\phantom{.......................}\\
\phantom{}\ce{\underset{(Alanine)}{CH3}}\phantom{..........................................}\ce{\underset{(Alanyl glycine)}{CH3}}\phantom{................}
\end{array}\]
Therefore, two dipeptide structures are possible from glycine and alanine.
They are
- glycyl alanine and
- Alanyl glycine
संबंधित प्रश्न
The chemical nature of peptide bond in proteins is ____________
Write types of proteins depending upon molecular shape.
Read the following statements and select the correct option.
- In quaternary structure, the peptide chains are much looped, twisted, and folded back on themselves due to formation of hydrogen bonds.
- When a protein has more than two polypeptide subunits their arrangement in space is called secondary structure.
Identify the option that correctly describes the amphoteric nature of proteins.
Identify the enzyme X in the following reaction.
\[\ce{H2O2(aq) ->[x] H2O(l) + \frac{1}{2} O2(g)}\]
An iron-containing red protein is ______
Which of the following amino acids has –CH2OH group in its side chain?
In a protein, various amino acids linked together by ____________.
Write the Zwitter ion structure of alanine.
Write the Zwitter ion structure of alanine.
