Advertisements
Advertisements
Question
1. \[\ce{A + H2O ->[ELECTROLYSIS] \underset{SODA}{\underset{CAUSTIC}{X}} + \underset{ANODE}{Y ^} + \underset{CATHODE}{Z ^}}\]
2. \[\ce{\underset{MOLTEN}{A} ->[ELECTROLYSIS] \underset{CATHODE}{M} + \underset{ANODE}{Y ^}}\]
3. \[\ce{M + CH3COOH -> P + Z ^}\]
4. \[\ce{CH3COOH + Q ->[Conc.][H2SO4] R}\]
5. \[\ce{R + X -> P + Q}\]
- Derive the names of A, Y, Z, M, P & R.
- Improvise an activity to test Z.
OR - Name the process in which compounds X, Y & Z are formed from A. Justify your response.
Activity
Derivation
Justify
Solution
- A - Sodium chloride
Y - Chlorine gas,
Z - Hydrogen gas,
M - Sodium metal,
P - Sodium ethanoate &
R - Ethyl ethanoate/ester - Any activity similar to the given figure:
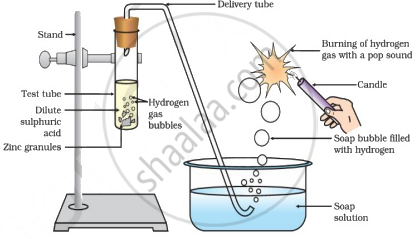
Reaction of zinc granules with dilute sulphuric acid and testing hydrogen gas by burning
OR - Chlor alkali process.
The process is called chlor alkali because of the products formed from chlorine and alkali for sodium hydroxide.
shaalaa.com
Is there an error in this question or solution?
