Advertisements
Advertisements
Question
A hydrocarbon ‘A’, \[\ce{(C4H8)}\] on reaction with \[\ce{HCl}\] gives a compound ‘B’, \[\ce{(C4H9Cl)}\], which on reaction with 1 mol of \[\ce{NH3}\] gives compound ‘C’, \[\ce{(C4H11N)}\]. On reacting with \[\ce{NaNO2}\] and \[\ce{HCl}\] followed by treatment with water, compound ‘C’ yields an optically active alcohol, ‘D’. Ozonolysis of ‘A’ gives 2 mols of acetaldehyde. Identify compounds ‘A’ to ‘D’. Explain the reactions involved.
Solution
\[\ce{(A) ->[Ozonolysis] 2CH3CHO}\]
\[\ce{\underset{(A)}{C4H8} ->[HCl] \underset{(B)}{C4H9Cl}}\]
Addition of \[\ce{HCl}\] has occurred on ‘A’. This implies ‘A’ is an alkene.
\[\ce{\underset{(B)}{C4H9Cl} -> \underset{(C)}{C4H11N}}\]
\[\ce{Cl}\] in compound ‘B’ is substituted by \[\ce{NH2}\] to give ‘C’.
\[\ce{(C) ->[NaNO2/HCl][H2O] (D)}\]
‘C’ gives a diazonium salt with \[\ce{NaNO2/HCl}\] that liberates \[\ce{N2}\] to give optically active alcohol. This means that ‘C’ is an aliphatic amine. Number of carbon atoms in amine is same as in compound ‘A’.
Since products of ozonolysis of compound ‘A’ are \[\ce{CH3 - CH = O}\] and \[\ce{O = CH - CH3}\]. The compound ‘A’ is \[\ce{CH3 - CH = CH - CH3}\]
On the basis of structure of ‘A’ reactions can be explained as follows:
\[\begin{array}{cc}
\phantom{}\ce{CH3 - CH = CH - CH3 ->[HCl] CH3CH2 - CH - CH3}\phantom{.}\\
\phantom{..........}|\phantom{...........................}|\phantom{......}\\
\phantom{......}\ce{\underset{(A)}{Cl}}\phantom{.........................}\ce{\underset{(B)}{Cl}}\phantom{.}\\
\end{array}\]
\[\begin{array}{cc}
\phantom{.}\ce{CH3 - CH2 - CH - CH3 ->[NH3] CH3 - CH2 - CH - CH3}\phantom{..}\\
\phantom{...............}|\phantom{..............................}|\phantom{...........}\\
\phantom{........}\ce{\underset{(B)}{Cl}}\phantom{............................}\ce{\underset{(C)}{NH2}}\phantom{..}\\
\end{array}\]
\[\begin{array}{cc}
\phantom{............................................}\ce{CH2CH3}\\
\phantom{.....................................}|\\
\phantom{.}\ce{CH3 - CH2 - CH - CH3 ->[NaNO2/HCl][H2O] CH3 - C - H}\phantom{..}\\
\phantom{...............}|\phantom{...................................}|\phantom{..............}\\
\phantom{..........}\ce{\underset{(C)}{NH2}}\phantom{...........................}\ce{\underset{(D)}{\underset{(Optically active)}{OH}}}\phantom{..}\\
\end{array}\]
APPEARS IN
RELATED QUESTIONS
Write the major products(s) in the following:
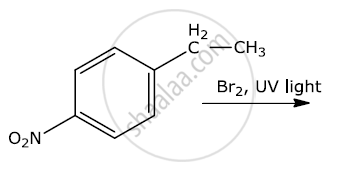
Write a note on Sandmeyer's reaction.
What is the action of benzene diazonium chloride on phenol in alkaline medium
What is the action of benzene diazonium chloride on Aniline
An aromatic compound 'A' on heating with Br2 and KOH forms a compound 'B' of molecular formula C6H7N which on reacting with CHCl3 and alcoholic KOH produces a foul-smelling compound 'C'. Write the structures and IUPAC names of compounds A, B, and C.
A compound with the molecular formula C5H5N and having 3 double bonds will be
Diazo-coupling is useful to prepare ______.
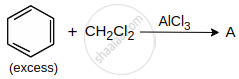
A is:
Write a structure of 2-Iodo-3 - methylpentane.
