Advertisements
Advertisements
Question
A radioactive substance is oxidized. Will there be any change in the nature its radioactivity? Give a reason for your answer
Solution
There will be no change in the nature of radioactivity of the substance. This is because oxidation is a chemical process and does not involve the nucleus of the substance.
APPEARS IN
RELATED QUESTIONS
State three factors on which the rate of emission of electrons from a metal surface depends
How can they be made to leave the metal surface? (State any two ways)
A radioactive source emits three types of radiations. Name them.
(i) Name the radiations which are charged.
(ii) Name the radiations which are most penetrating.
(iii) Name the radiations which travel with the speed of light.
(iv) Name the radiations which have the largest mass.
In the following fig. shows a radioactive source S in a thick lead container. The radiations pass through an electric field between the plates P and Q. Complete the diagram to show the paths of α , β and γ radiations.
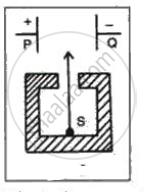
Why is the source S placed in a thick lead container?
A radioactive nuclide `""_86^226` Ra decays by emission of two alpha particles, one beta particle and gamma rays. Which of the following is the resulting nuclide X?
Name two radioactive substances.
It is advised not to touch a radioactive substance by hand. Give reason.
Unit of radioactivity is _______
A radioactive nucleus \[\ce{^A_ZX}\] undergoes spontaneous decay in the sequence \[\ce{^A_ZX -> _{Z-1 }B -> _{Z-3}C -> _{Z-2}D }\], where Z is the atomic number of element X. The possible decay particles in the sequence are ______.
