Advertisements
Advertisements
Question
A reddish-brown metal ‘X’, when heated in air, gives a black compound ‘Y’, which when heated in the presence of H2 gas gives ‘X’ back. ‘X’ is refined by the process of electrolysis; this refined form of ‘X’ is used in electrical wiring. Identify ‘X’ and ‘Y’. Draw a well-labelled diagram to represent the process of refining ‘X’.
Solution
- ‘X’ - Copper/Cu and ‘Y’ - CuO.
- The diagram represents the process of refining ‘X'.
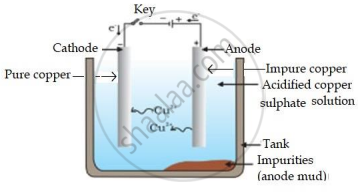
APPEARS IN
RELATED QUESTIONS
What is aqua-regia? Name two special metals which are insoluble in common reagents but dissolve in aqua-regia.
Name any five metals which are purified by electrolytic refining method.
During the refining of an impure metal by electrolysis, the pure metal is a deposited:
(a) at cathode
(b) on the walls of electrolytic tank
(c) at anode
(d) at the bottom of electrolytic tank
Draw a neat labelled diagram.
Magnetic separation method.
Which of the following process to be carried out to avoid the
formation of greenish layer on brass vessels due to corrosion?
Name one metal each occurring as :
A sulphine
\[\ce{Cu2S + 3Cu2O -> 6Cu + SO2}\]
The above process is:
Bauxite is mixed with cryolite so as to
(i) reduce its melting point
(ii) increase its electrical conductivity
(iii) molten cryolite acts as a solvent
(iv) increase its melting point
Identify the terms for the following:
The electrode where reduction takes place.
Answer the following questions with reference to the electrorefining of copper:
Write the reaction taking place at the cathode.
