Advertisements
Advertisements
Question
A schematic plot of ln Keq versus inverse of temperature for a reaction is shown below
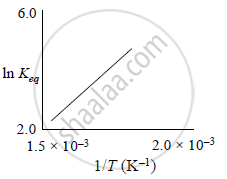
The reaction must be:
Options
highly spontaneous at ordinary temperature
one with negligible enthalpy change
Endothermic
Exothermic
MCQ
Solution
Exothermic
Explanation:
`"K"_"f" = "A"_"f" e^(-"E"_"f"//"RT") and "K"_"b" = "A"_"b" e^(- "E"_"b"//"RT")`
We know that `"K"_"eq" = "K"_"f"/"K"_"b" => "K"_"eq" = "A"_"f"/"A"_"b" e^(- ("E"_"f" - "E"_"b")//"RT")`
`=> ln "K"_"eq" = ln "A"_"f"/"A"_"b" - ("E"_"f" - "E"_"b")/"RT"`
`=> ln "K"_"eq" = (- Delta "H")/"R" (1/"T") + ln "A"_"f"/"A"_"b"`
Comparing this equation with y = mx + c
m = `(- Delta "H")/"R" = + "ve"`
Thus, ΔH < 0 or reaction is exothermic.
shaalaa.com
Is there an error in this question or solution?
