Advertisements
Advertisements
Question
A solution of phenol in chloroform when treated with aqueous NaOH gives compound P as a major product. The mass percentage of carbon in P is ______. (to the nearest integer) (Atomic mass: C = 12; H = 1; O = 16)
Options
75
69
56
30
MCQ
Fill in the Blanks
Solution
A solution of phenol in chloroform when treated with aqueous NaOH gives compound P as a major product. The mass percentage of carbon in P is 69.
Explanation:
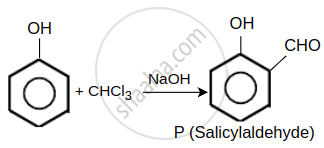
Molecular formula of product 'P' = C7H6O2
So, mass % of C in 'P' = `(12 xx 7)/(84 + 6 + 32) xx 100 = 68.85% ~~ 69%`
shaalaa.com
Is there an error in this question or solution?
