Advertisements
Advertisements
Question
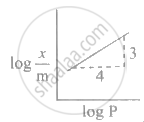
Adsorption of a gas follows Freundlich adsorption isotherm. x is the mass of the gas adsorbed on mass m of the adsorbent. The plot of log `"x"/"m"`· versus log P is shown in the given m graph. `"x"/"m"` is proportional to
Options
P4
P3
`"P"^(4/3)`
`"P"^(3/4)`
MCQ
Solution
`"P"^(3/4)`
Explanation:
According to the Freundlich adsorption isotherm:
`"x"/"m" ∝ "P"^(1/"n")`
∴ `log "x"/"m" = log "k" + 1/"n" log "P"`
This corresponds to the equation of a straight line: y = mx + c
∴ Slope of the line (m) is equal to `1/"n" = 3/4`
∴ `"x"/"m" ∝ "P"^(3/4)`
shaalaa.com
Is there an error in this question or solution?
