Advertisements
Advertisements
Question
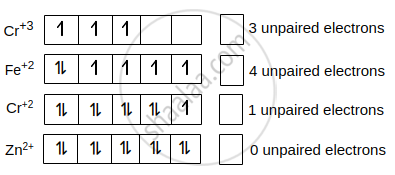
Among the following ions, which one has the highest paramagnetism?
Options
\[\ce{[Cr(H2O)6]^{3+}}\]
\[\ce{[Fe(H2O)6]^{2+}}\]
\[\ce{[Cu(H2O)6]^{2+}}\]
\[\ce{[Zn(H2O)6]^{2+}}\]
MCQ
Solution
\[\ce{[Fe(H2O)6]^{2+}}\]
Explanation:
The compound with highest no. of unpaired electrons has highest paramagnetism.
shaalaa.com
Is there an error in this question or solution?
