Advertisements
Advertisements
Question
An aldehyde isomeric with allyl alcohol gives phenyl hydrazone. Pick out a ketone that too gives a phenyl hydrazone containing the same percentage of nitrogen.
Options
Methyl ethyl ketone
Dimethyl ketone
2-Butanone
2-Methyl propanone
MCQ
Solution
Dimethyl ketone
Explanation:
\[\ce{\underset{Allyl alcohol}{CH2 = CH - CH2 - OH} -> \underset{\underset{(Propanol)}{↓\phantom{......}}}{CH3}CH2 - CHO}\]
Propanole is an isomer of Allyl alcohol.
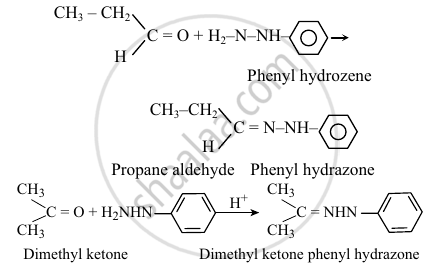
Both hydrazones contained the same percentage of Nitrogen.
shaalaa.com
Is there an error in this question or solution?
