Advertisements
Advertisements
Question
An element belongs to group II. What would be its valency? Write the formula of molcules of compounds it will form with elements in VA, VIA and VIIA groups.
Short Note
Solution
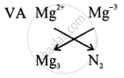
VIA
| Symbols | Valencies | ||
| Magnesium | Oxide | Magnesium | Oxide |
| Mg | O | 2+ | 2- |
(1) Write the symbol
| On the left-hand side Calcium | On the right-hand side Chloride |
| Ca | Cl |
(2) Write the valency of the symbols
| At the top right corner Positive ion |
At the top right corner Negative ion |
| Ca2+ | Cl1- |
(3) Interchange the valency
number (Ignore the (+)and (-) signs)
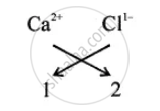
(4) Write the interchanged numbers at the base
but ignore the + and the - sings
`"Ca"_1` `"Cl"_2`
shaalaa.com
Is there an error in this question or solution?
