Advertisements
Advertisements
Question
An ideal gas is compressed to half its initial volume by means of several processes. Which of the process results in the maximum work done on the gas?
Options
Isothermal
Adiabatic
Isobaric
Isochoric
MCQ
Solution
Adiabatic
Explanation:
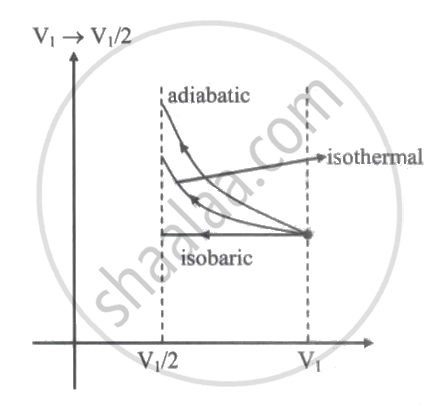
Work done = area under curve Wadiabatic > Wisothermal > Wisobaric For isochoric process, W = 0
shaalaa.com
Thermodynamic Process
Is there an error in this question or solution?
