Advertisements
Advertisements
Question
Answer the following in brief.
What are the requirements for the colliding reactant molecules to lead to products?
Solution
The requirements for colliding molecules to form the product are as follows:
i. Activation energy: For the reaction to occur the colliding reactant molecules must possess the minimum kinetic energy. This minimum kinetic energy is the activation energy. The reaction would occur only if colliding molecules possess kinetic energies equal to or greater than the activation energy.
ii. The orientation of reactant molecules:
a. For reactions involving complex molecules or ions, reactant molecules must collide with proper orientation. The molecules need to be so oriented relative to each other that the reacting groups approach closely.
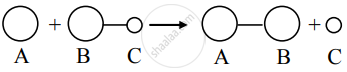
b. Consider, A + C - B → A - B + C
The collision of A with C approaching toward A would not lead to a reaction.

The reactant molecules would collide and separate owing to the improper orientation of C – B.
c. The reaction is successful as a result of proper orientation of C – B. A fraction of such collisions brings forth the conversion of reactants to products.
iii. Potential energy barrier:
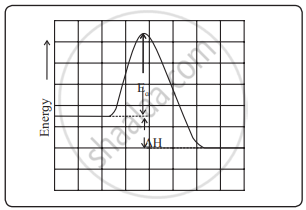
Consider the reaction A + C – B → A – B + C
a. During a course of the collision, new bond A – B develops, and at the same time bond C – B breaks. An activated complex is formed in which all three atoms are weakly connected together. A + C – B → A-----B-----C → A – B + C
b. To attain the configuration A-----B-----C atoms need to gain energy, which comes from the kinetic energy of colliding molecules. The energy barrier between reactants and products is as shown in the diagram.
Potential energy barrier
c. To form an activated complex, the reactant molecules need to climb up and overcome the energy barrier before they get converted to products.
d. The height of the barrier is called activation energy (Ea). Thus, the reactant molecules transform into products only if they possess energy equal to or greater than such activation energy. As a result, only a few collisions lead to products.
APPEARS IN
RELATED QUESTIONS
Answer the following in one or two sentences.
Why all collisions between reactant molecules do not lead to a chemical reaction?
Answer the following in one or two sentences
What is the activation energy of a reaction?
Effect of catalyst in a chemical reaction is to change the _______.
The potential energy of the transition state is ____________.
