Advertisements
Advertisements
Question
Assertion: When water is heated up to 100°C, there is no raise in temperature until all water gets converted into water vapour.
Reason: Boiling point of water is 10°C.
Options
If both assertion and reason are true and reason is the correct explanation of assertion.
If both assertion and reason are true but reason is not the correct explanation of assertion.
If assertion is true but reason is false.
If assertion is false but reason is true.
Solution
If assertion is true but reason is false.
APPEARS IN
RELATED QUESTIONS
State the three units of temperature.
Convert 40°C to the (a) Fahrenheit scale (b) Kelvin Scale.
Convert – 40°F to the Celsius scale.
Two substances A and B have specific heats c and 2 c respectively. If A and B are given Q and 4Q amounts of heat respectively, the change in their temperatures is the same. If the mass of A is m, what is the mass of B?
Distinguish between heat and temperature.
S.I. unit of temperature is :
Express Kelvin Zero in °C
Define heat energy on the basis of kinetic theory of matter.
What is the principle of calorimetry?
Name three devices which are used to detect heat radiations.
A small gap is left between two iron rails of the railway track. Why?
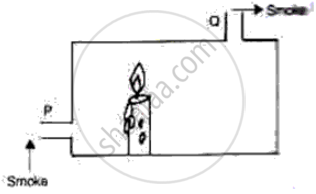
The following figure shows a container in which a lighted candle has been kept. The container has two openings, p, and Q. Which way the smoke from an external source placed near P moves when it reaches P and leaves from Q. Give a reason for your answer.
Heat flows from a ______ body to a _____ body.
Paheli and Boojho measured their body temperature. Paheli found her’s to be 98.6 °F and Boojho recorded 37°C. Which of the following statement is true?
