Advertisements
Advertisements
Question
Briefly describe the structure of an atom.
Solution
STRUCTURE OF ATOM:

Atom: has an equal number of +ve charge (protons) and -ve charge electrons and hence atom has no charge.
Neutrons (having no charge) and protons are present in the nucleus. Electrons keep revolving around the nucleus in different shells.
APPEARS IN
RELATED QUESTIONS
If a glass rod rubbed with silk is brought near the cap of a negatively charged electroscope, the divergence of leaves will decrease.
In induction, a positively charged body can make an uncharged body positively charged.
When an ebonite rod is rubbed with fur, the rod acquires
Charging a conductor by bringing another charged conductor close to it without touching is called
The factor responsible for charging a conductor is
Name two substances which can be charged by friction.
Name three constituents of an atom and state the kind of charge on each of them.
What is the net charge on an atom?
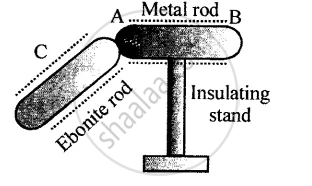
Figure below shows a metal rod AB placed on an insulating stand.
In figure (a) a negatively charged ebonite rod C is touched with the metal rod AB, while in figure (b), the negatively charged ebonite rod C is held near the rod AB. State the kind of charges at the ends A and B of the rod, in each case.
How will you use a gold leaf electroscope to find out whether the charge on a charged body is positive or negative?
