Advertisements
Advertisements
Question
Calculate the number of molecules of sulphur (S8) present in 16 g of solid sulphur.
Solution
1 mole of solid sulphur (S8) = 8 × 32 g = 256 g
i.e., 256 g of solid sulphur contains = 6.022 × 1023 molecules
`"Then, 16 g of solid sulphur contains "=(6.022xx10^(23))/256xx16" molecules"`
= 3.76 × 1022 molecules (approx)
APPEARS IN
RELATED QUESTIONS
Write down the name of a compound represented by the following formula:
Al2(SO4)3
Write down the name of a compound represented by the following formula:
KNO3
Give the name of the element present in the following compound:
Quick lime
Give the name of the element present in the following compound:
Potassium sulphate
Define: Mass number :
Write the formulae of the following compounds. Also name the elements present in them.
- Water
- Ammonia
- Methane
- Sulphur dioxide
- Ethanol
An element A forms an oxide A2O5.
What is the valency of element A ?
State the atomicity of the following:
Argon
Classify the following molecules as the molecules of element or compound
| 1. |  |
|
| 2. |  |
|
| 3. | 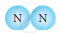 |
|
| 4. | 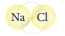 |
Which of the following contains maximum number of molecules?
