Advertisements
Advertisements
Question
Calculate packing efficiency in face-centred cubic lattice.
Solution
Step 1: Radius of sphere:
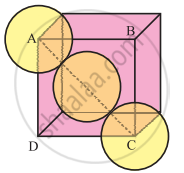
In the unit cell of face-centred cubic lattice, there 8 atoms at 8 corners and 6 atoms at 6 face centres.
Consider the face ABCD.
The atoms are in contact along the face diagonal BD. Let a be the edge length and r, the radius of an atom.
Consider a triangle BCD.
BD2 = BC2 + CD2
= a2 + a2 = 2a2
`∴ "BD" = sqrt2 a`
From figure, BD = 4r
`∴ 4r = sqrt2a and r - sqrt2/4a = a/(2sqrt2)`
Step 2: Volume of sphere:
`"Volume of one particle" = 4/3 πr^3`
`= (4π)/3 xx (a/(2sqrt2))^3`
`= 4/3πa^3(1/(2sqrt2))^3`
`= (πa^3)/(12sqrt2)`
`"total volume of 4 particles in unit cell" = 4 xx (πa^3)/(12sqrt2) = (πa^3)/(3sqrt2)`
Step 3: Packing efficiency:
`"Packing efficiency" = ("Volume occupied by particles in unit cell")/("Volume of unit cell")xx 100`
`= ((πa^3)/(3sqrt2))/a^3 xx 100`
`= (3.142)/(3sqrt2) xx 100 = 74%`
