Advertisements
Advertisements
Question
Classify the following compound as a primary, secondary and tertiary halide.
1-Bromobut-2-ene
Options
Primary halide
Secondary halide
Tertiary halide
Solution
Primary halide
Explanation:
\[\ce{\underset{1-Bromobut-2-ene}{CH3HC = CHCH2Br}}\]
APPEARS IN
RELATED QUESTIONS
Draw the structures of major monohalo products in each of the following reaction:
Name the following halide according to the IUPAC system and classify it as an alkyl, allyl, benzoyl (primary, secondary, tertiary), vinyl or aryl halide:
CH3C(Cl)(C2H5)CH2CH3
Name the following halide according to the IUPAC system and classify it as an alkyl, allyl, benzoyl (primary, secondary, tertiary), vinyl or aryl halide:
CH3CH=CHC(Br)(CH3)2
Name the following halide according to the IUPAC system and classify it as an alkyl, allyl, benzoyl (primary, secondary, tertiary), vinyl or aryl halide:
m-ClCH2C6H4CH2C(CH3)3
m-Xylene reacts with Br2 in presence of FeBr3, what are products formed:
Which of the following compounds can be classified as aryl halides?
(i) \[\ce{p-ClC6H4CH2CH(CH3)2}\]
(ii) \[\ce{p-CH3CHCl(C6H4)CH2CH3}\]
(iii) \[\ce{o-BrH2C - C6H4CH(CH3)CH2CH3}\]
(iv) \[\ce{C6H5 - Cl}\]
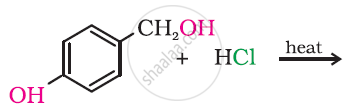
Why can aryl halides not be prepared by reaction of phenol with \[\ce{HCl}\] in the presence of \[\ce{ZnCl2}\]?
Two isomers (A) and (B) with molar mass 184 g/mol and elemental composition C 52.2%; H 4.9% and Br 42.9% gave benzoic acid and p-bromobenzoic acid, respectively on oxidation with KMnO4. Isomer ‘A’ is optically active and gives a pale yellow precipitate when warmed with alcoholic AgNO3. Isomer ‘A’ and ‘B’ are, respectively:
Name the following halide according to the IUPAC system and classify them as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl, or aryl halide:
\[\ce{CH3CH2C(CH3)2CH2I}\]
Name the following halide according to IUPAC system and classify them as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halide:
\[\ce{CH3C(CI)(C2H5)CH2CH3}\]
