Advertisements
Advertisements
Question
Classify the following solutions as acids, bases or salts.
Ammonium hydroxide, barium chloride, sodium chloride, sodium hydroxide, H2SO4 and HNO3
Solution
Acids: H2SO4 and HNO3
Bases: Ammonium hydroxide and sodium hydroxide.
Salts: Barium chloride and sodium chloride.
APPEARS IN
RELATED QUESTIONS
How will you test for the presence of this gas?
What are organic acids and mineral acids?
A white shirt has a yellow stain of curry. When soap is rubbed on this shirt during washing, the yellow stain turns reddish-brown. On rinsing the shirt with plenty of water, the reddish-brown stain turns yellow again.
Explain the changes in colour of this indicator which take place during washing and rinsing the shirt.
Wasp sting contains:
(a) a sugar solution
(b) an acidic liquid
(c) a salt solution
(d) an alkaline liquid
The label on the bottle of chemical is spoiled. How will you find whether the chemical is acidic or not?
Match the pairs.
| Group A | Group B |
| 1. Tamarind | a. Acetic acid |
| 2. Curd | b. Citric acid |
| 3. Lemon | c. Tartaric acid |
| 4. Vinegar | d. Lactic acid |
In acidic solutions blue litmus changes into ______ colour.
Which of the following is an acid-base indicator?
Fill in the crossword given in Figure 5.2 with the help of the clues provided.
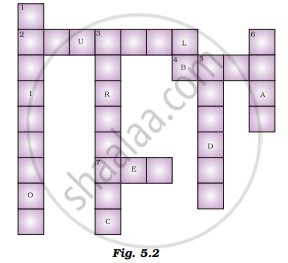
Across
(2) The solution which does not change the colour of either red or blue litmus.
(4) Phenolphthalein gives pink colour in this type of solution.
(7) Colour of blue litmus in lemon juice.
Down
(1) It is used to test whether a substance is acidic or basic.
(3) It is a natural indicator and gives pink colour to the basic solution.
(5) Nature of ant’s sting.
(6) It is responsible for the increase in temperature during a neutralisation reaction.
The acid which contains four hydrogen atoms ______.
