Advertisements
Advertisements
Question
Compare the structures of H2O and H2O2.
Solution
Both in gas-phase and liquid-phase, the molecule adopts a skew confirmation due to repulsive interaction of the OH bonds with lone pairs of electrons on each oxygen atom. Indeed, it is the smallest molecule known to show hindered rotation about a single bond.
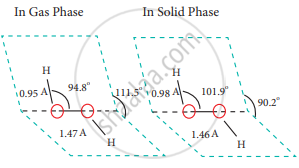
H2O2 has a non-polar structure. The molecular dimensions in the gas phase and solid phase differ as shown in the figure. Structurally, H2O2 is represented by the dihydroxyl formula in which the two OH-groups do not lie in the same plane.
One way of explaining the shape of hydrogen peroxide is that the hydrogen atoms would lie on the pages of a partly opened book, and the oxygen atoms along the spine. In the solid phase of the molecule, the dihedral angle reduces to 90.2° due to hydrogen bonding and the O-O-H angle expands from 94.8° to 101.9°. Water has a bent structure and the H-O-H bond angle is 104.5°.
APPEARS IN
RELATED QUESTIONS
Give balanced chemical equations for Hydrogen peroxide is treated with an acidified KMnO4 solution.
Give balanced equation for the following reaction:
Lead sulphide is heated with hydrogen peroxide.
When hydrogen peroxide is shaken with an acidified solution of potassium dichromate in presence of ether, the ethereal layer turns blue due to the formation of
Hydrogen peroxide can function as an oxidising agent as well as the reducing agent. substantiate this statement with suitable examples.
