Advertisements
Advertisements
Question
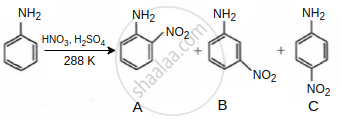
Consider the given reaction, percentage yield of:
Options
C > B > A
C > A > B
B > C > A
A > C > B
MCQ
Solution
C > B > A
Explanation:
During aniline nitration in a strong acidic (HNO3, H2SO4) medium, aniline transforms into anilinium ion, which can withdraw electron density. Its effect is strongest at the ortho, then meta, and finally para. As a result, very little ortho-nitrated product is formed.
shaalaa.com
Electrophilic Aromatic Substitution in Aromatic Amines
Is there an error in this question or solution?
