Advertisements
Advertisements
Question
Describe the bonding in the tetrahedral complex Ni(CO)4 on the basis of valence bond theory. Give the orbital diagrams of metal atoms in free state and in the complex. Mention the number of unpaired electrons in the complex.
Answer in Brief
Solution
Ni (CO)4 (tetrahedral), oxidation state of Ni is zero.
Electron configuration of Ni: [Ar]3d84s2
Orbital diagram of free metal atom:

The complex is tetrahedral. Ni must be sp3 hybridized. Therefore, in the presence of ligands, 3d electrons are forced to pair up and two 4s electrons are transferred to vacant d-orbital. One 4s and three 4p orbitals hybridize to form four sp3 vacant hybrid orbitals which overlap with filled orbitals of ligands to form coordinate bonds.
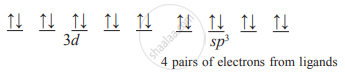
∴ number of unpaired electrons = 0.
shaalaa.com
Theories of Bonding in Complexes
Is there an error in this question or solution?
