Advertisements
Advertisements
Question
Differentiate between metals and non-metals with reference to –
(a) number of electrons in outer or valence shell
(b) formation of cation and anion
(c) reaction with dilute acids.
Solution
| Chemical Property | Metals | Non-metals |
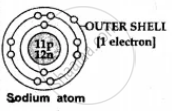
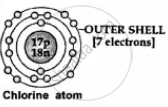
| (a) No.of electrons in outer shell |
1, 2 or 3 electrons -
|
4, 5, 6 or 7 electrons - in the outer or valence shell of a non-metal
|
| (b) Ion formation | Metal lose or donate valence electrons and form cations [positive ions] e.g. Na - 1e- → Na1+ [caution] |
Non-metal accept valence electrons and form anions [negative ions] e.g. Cl + 1e- → Cl1- [anion] |
| (c) Reaction with acids | React with dilute acids to give hydrogen | Do not react with dilute acids to give hydrogen. |
APPEARS IN
RELATED QUESTIONS
Name two non-metals which are both brittle and non-ductile.
"Is malleable and ductile". This best describes:
(a) a metal
(b) a compound
(c) a non-metal
(d) a solution
Which of the following alloys contains a non-metal as one of the constituents?
(a) brass
(b) amalgam
(c) steel
(d) bronze
Name a metal as precious as gold
Name a metal used as a thermometric liquid
Name a non-metal that is used for filling into electric bulbs
Statements given below are incorrect. Write the correct statements :
Chlorine gas is used in the manufacture of sulphuric acid.
What do you understand by the term non-metal?
Name the non-metal present in abundance in the earth's crust.
Name:
a metal with dull appearance
Name:
a metal which can be cut with a knife.
Select from the following ist:
`Fe_2O_3, , NO, PbO, Mn_2O_7`
Amphoteric oxide …………
Name:
two non metals which are monoatomic
Name a non-metal which shows reducing property.
Name:
a metal whose oxide is reduced only by carbon
Give the effect of heat on metal oxides based on the activity series ?
Which metal occurs as a sulphide also gives the name of their respective ore.
Gold and silver are used to make jewellery. Give reason
Give a reason why – properties of metals are related to their activity or reactivity series of metals. Explain the same with reference to potassium and iron in the series.