Advertisements
Advertisements
Question
Discuss the three states of matter solid, liquids and gas on the basis of molecular model.
Solution
Solids

Here the molecules are very tightly packed that there is no or very less intermolecular space and there is a high intermolecular force of attraction (force of cohesion). The molecules in a solid can only vibrate to and fro about their mean positions. They do not leave their positions.
Liquids
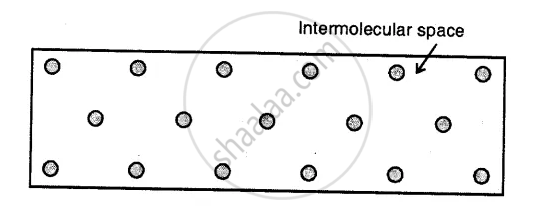
Here the molecules are less tightly packed as compared to solids and also there is a lesser force of intermolecular attraction. The intermolecular distance is greater than that in the solids. Thus, they do not have a definite shape but acquire the shape of the vessel in which they are contained but have a definite volume at a given temperature.
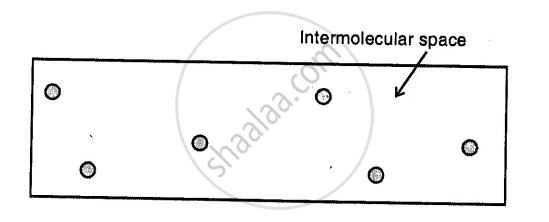
Gases :
APPEARS IN
RELATED QUESTIONS
Water at 100°C has more heat than the steam at 100°C.
Write true or false for the following statement
The liquids are less viscous than the gases.
Fill in the blank
The gases are .......... dense.
How do the solids, liquids, and gases differ in their following properties.
(a) Size
(b) Shape
(c) Density
Differentiate between the following:
Liquids and gases
Fill in the blank
......... can flow.
For the following statement, say whether it describes a solid, a liquid, or a gas.
Particles move about very quickly.
Multiple choice question.
The state of matter which has no definite shape or volume is called
Multiple choice question.
Gases
State the correct term from A, B, C, D, E, or F in List II which represents the change of state of matter or its relevant property from List I.
| List I | List II |
| 1. Solid 'X' to a Liquid 'X' | A: Condensation |
| 2. Liquid 'Y' to its vapour 'Z' | B: Vaporization |
| 3. 'Z' to 'Y' | C: Melting |
| 4. 'Y' to 'X' | D: Freezing |
| 5. The temperature at which 'Y' changes to 'Z' | E: Melting point |
| F: Boiling point |
