Advertisements
Advertisements
Question
Do you expect the carbon hydrides of the type (CnH2n + 2) to act as ‘Lewis’ acid or base? Justify your answer.
Solution
For carbon hydrides of type CnH2n + 2, the following hydrides are possible for
`"n" = 1 => "CH"_4`
`"n" = 2 => "C"_2"H"_6`
`"n" = 3 => "C"_3"H"_8`
For a hydride to act as a Lewis acid i.e., electron-accepting, it should be electron-deficient. Also, for it to act as a Lewis base i.e., electron donating, it should be electron-rich.
Taking C2H6 as an example, the total number of electrons is 14 and the total covalent bonds are seven. Hence, the bonds are regular 2e–-2 centered bonds.
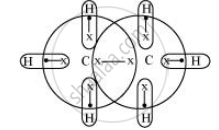
Hence, hydride C2H6 has sufficient electrons to be represented by a conventional Lewis structure. Therefore, it is an electron-precise hydride, having all atoms with complete octets. Thus, it can neither donate nor accept electrons to act as a Lewis acid or Lewis base.
APPEARS IN
RELATED QUESTIONS
What do you understand by electron-rich compounds of hydrogen? Provide justification with a suitable example.
What characteristics do you expect from an electron-deficient hydride with respect to its structure and chemical reactions?
Arrange the following:
LiH, NaH and CsH in order of increasing ionic character.
What do you understand by electron-deficient? Provide justification with a suitable example.
What do you understand by electron-precise? Provide justification with a suitable example.
Name the classes of hydrides to which \[\ce{H2O, B2H6}\] and \[\ce{NaH}\] belong.
What do you understand by the term ‘auto protolysis of water’ ? What is its significance?
Molecular hydrides are classified as electron-deficient, electron precise and electron-rich compounds. Explain each type with two examples.
Hydrogen generally forms covalent compounds. Give reason.
An ionic hydride of an alkali metal has significant covalent character and is almost unreactive towards oxygen and chlorine. This is used in the synthesis of other useful hydrides. Write the formula of this hydride. Write its reaction with \[\ce{Al2Cl6}\].
