Advertisements
Advertisements
Question
Draw the structure of BeCl2 (vapour).
Solution
BeCl2 (vapour)
In the vapour state, it exists as a chlorobridged dimer.
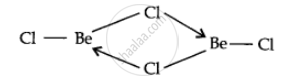
APPEARS IN
RELATED QUESTIONS
Draw the structure of BeCl2 (solid).
Which of the carbonates given below is unstable in air and is kept in CO2 atmosphere to avoid decomposition.
Present a comparative account of the alkali and alkaline earth metals with respect to the following characteristics:
(i) Tendency to form ionic/covalent compounds
(ii) Nature of oxides and their solubility in water
(iii) Formation of oxosalts
(iv) Solubility of oxosalts
(v) Thermal stability of oxosalts
Which of the following alkaline earth metal sulphates is least soluble in water?
The structures of beryllium chloride in the solid state and vapour phase, are ______.
Given below are two statements:
Statement I: Both CaCl2.6H2O and MgCl2.8H2O undergo dehydration on heating.
Statement II: BeO is amphoteric whereas the oxides of other elements in the same group are acidic.
In the light of the above statements, choose the correct answer from the options given below:
An inorganic salt solution gives a yellow precipitate with silver nitrate. The precipitate dissolves in dilute nitric acid as well as in ammonium hydroxide. The solution contains _______.
\[\ce{BaC2 + N2 ->[\Delta] (1)}\]
\[\ce{CaC2 + N2 ->[\Delta] (2)}\] (1) and (2) are ______.
Ca, Ba and Sr ions are precipitated in fifth group as their ______.
Halides of alkaline earth metals form hydrate such as MgCl2.6H2O, CaCl2.6H2O, BaCl2.2H2O and SrCl2.2H2O. This shows that halides of group 2 elements:
Number of amphoteric compound among the following.
