Advertisements
Advertisements
Question
During summer days, water kept in an earthen pot (pitcher) becomes cool because of the phenomenon of :
Options
diffusion
transpiration
osmosis
evaporation
Solution
evaporation
An earthen pot has a large number of extremely small pores on its wall. Some of the water continuously keeps seeping through these pores. This water evaporates continuously and takes the latent heat required for vaporization from the earthen pot and the remaining water. In this way, the remaining water loses heat and gets cooled.
APPEARS IN
RELATED QUESTIONS
Why is cooling produced on evaporation of a liquid?
A patient suffering from high fever is advised to put wet clot strips on his forehead. Why?
Would you cool a bucket of water more quickly by placing it on ice or by placing ice in it ? Give reasons for your answer.
Which produces more severe burns : boiling water or steam ? Why ?
Why does evaporation cool a liquid ?
Why is the air in a region dry?
Look at Fig. 1.3 and suggest in which of the vessels A,B, C or D the rate of evaporation will be the highest? Explain.
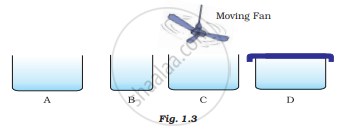
Comment on the following statements:
Evaporation produces cooling.
The rate of evaporation is more when the surface area is greater.
When lemon juice is mixed with soda water, they produce brisk effervescence.
