Advertisements
Advertisements
Question
Electrophilic addition reactions proceed in two steps. The first step involves the addition of an electrophile. Name the type of intermediate formed in the first step of the following addition reaction.
\[\ce{H3C - HC = CH2 + H+ ->?}\]
Options
2° Carbanion
1° Carbocation
2° Carbocation
1° Carbanion
MCQ
Solution
2° Carbocation
Explanation:
When the electrophile attacks CH3 – CH = CH2, delocalisation of electrons can take place in two possible ways.
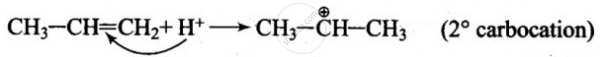
\[\ce{CH3 - CH2 - \overset{⊕}{C}H2}\] (1° carbanion)
As 2° carbocation is more stable than 1° carbocation, the first addition is more feasible.
shaalaa.com
Fundamental Concepts in Organic Reaction Mechanism - Types of Organic Reactions and Mechanisms
Is there an error in this question or solution?
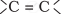 portion which can be shown as:
portion which can be shown as: