Advertisements
Advertisements
Question
Elements X and Y have the following configurations:
X (2, 8, 7), Y (2, 8, 8, 2)
Answer the following questions:
(a) What will be the nature of bond between X and Y?
(b) Draw the diagram to show the bond formation between X and Y?
(c) Sate three main properties of this compound.
Solution
Electronic Configuration of X shows that X is a metal.
Electronic Configuration of Y shows that y is a non metal.
a) Since Y is a metal and thus electropositive :
Y → Y+ + 2e-
B) Since X is a non metal and thus electro-negative :
X + 1e- → X-
Hence two X atoms can combine with one Y atom to form YX2 molecule and since the formation of YX2 involves complete transfer of electrons between them so the bond between them is ionic.
(b) 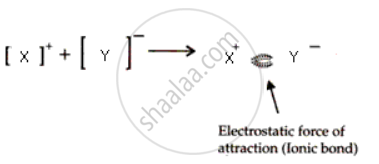
(c) The three main properties of this compound are:
- It is an ionic compound.
- It is soluble in water but insoluble in organic solvents.
- It is a good conductor of electricity in the molten state and in aqueous solution.
