Advertisements
Advertisements
Question
Energy levels A, B, C of acertain atom corresponding to increasing value of energy, i.e., EA< E8 < Ee. If λ1, λ2 and λ3 are the wavelength of radiations corresponding to the transitions C to B, B to A and C to A respectively, which of the following statements is correct?
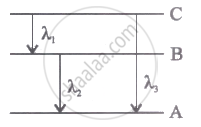
Options
λ3 = λ1 + λ2
λ1 = `(lambda_1lambda_2)/(lambda_1 + lambda_2)`
λ1 + λ2 + λ3 = 0
`lambda_3 ^2 = lambda_1 ^2 + lambda_2^2`
MCQ
Solution
λ1 = `(lambda_1lambda_2)/(lambda_1 + lambda_2)`
Explanation:
(EC - EB) + (EB - EA) = EC - EA
`Delta "E"_"CB" + Delta "E"_"BA" = Delta "E"_"CA"`
As we know,
`Delta "E"_"a" = "hc"/lambda_"n"` then,
`"hc"/lambda_1 + "hc"/lambda_2 = "hc"/lambda_3`
`1/lambda_1 + 1/lambda_2 = 1/lambda_3`
`=> lambda_3 = (lambda_1 lambda_2)/(lambda_1 + lambda_2)`
shaalaa.com
Is there an error in this question or solution?
