Advertisements
Advertisements
Question
explain catalytic oxidation of ammonia.
Solution
In the presence of Platinum at 800°C, ammonia reacts with oxygen to give nitric oxide and water vapour.
Procedure:
Pass dry ammonia gas and oxygen gas through inlets over heated platinum placed in the combustion tube, which in the heated state emits reddish glow.
Reaction:
4NH3+ 5O2 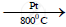 4NO + 6H2O + Heat
4NO + 6H2O + Heat
2NO + O2 → 2NO2
Brown gas
Observation:
Reddish brown vapours of nitrogen dioxide are seen in the flask due to the oxidation of nitric oxide.
The platinum continues to glow even after the heating is discontinued since the catalytic oxidation of ammonia is exothermic.
shaalaa.com
Chemical Properties of Ammonia
Is there an error in this question or solution?
