Advertisements
Advertisements
Question
Explain in brief – Lod Rutherford’s experiment which led to the discovery of the atomic nucleus.
Solution
Discovery of – Atomic nucleus:
- Discovery – Study of the atomic model leading to the discovery of – atomic nucleus.
Experiment:
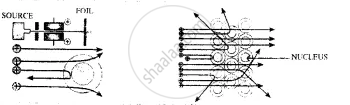
- Rutherford projected alpha particles towards a thin gold foil, – in the path of the rays.
- He saw that most of the alpha particles went straight through the foil, – but some were deflected slightly & some by large angles.
Conclusion:
- An atom, on the whole, is relatively empty but consists of a – concentrated positive mass in the centre, which leads to the deflection of the alpha particles.
- Thus the discovery of a central positive region – atomic nucleus was initiated.
APPEARS IN
RELATED QUESTIONS
Multiple Choice Questions
The number of electrons in an atom is equal to number of
Draw the diagrams representing the atomic structures of the following: Nitrogen.
Tick the most appropriate answer
Which one of the following scientists put forward the theory regarding the extra-nuclear structure of the atom?
- James Chadwick
- John Dalton
- Rutherford
- Niels Bohr
ANSWER IN DETAIL
How are radioactive radiations useful in medicines ?
Which part of atom was discovered by Rutherford?
How was it shown that atom has empty space?
Where is the nucleus of an atom situated?
Explain in brief the experimental proof which led to the discovery of –
Atomic nucleus
What is meant by the term nucleus
Differentiate between the following term:
Nucleus and nucleons
