Advertisements
Advertisements
Question
Explain: Phenols are acid while alcohol is neutral.
Short Note
Solution
- Ionisation of alcohol:
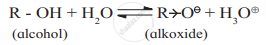
Electron donating inductive effect (+I effect) of alkyl group destabilizes the alkoxide ion (the conjugate base of alcohol). As a result alcohol does not ionize much in water, and behaves like neutral compound in aqueous medium. - Ionisation of phenol:
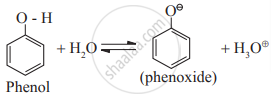
Phenoxide ion, the conjugate base of phenol, is resonace stabilized by delocalization of the negative charge.Therefore phenol ionizes in aqueous medium to a moderate extent, and thereby shows a weak acidic character.
shaalaa.com
Alcohols and Phenols
Is there an error in this question or solution?
