Advertisements
Advertisements
Question
Explain Sidgwick’s electronic theory with suitable example
Solution
The Sidgwick’s electronic theory explains the formation of coordination compounds. According to this theory, coordinate bonds are formed when the ligands donate the electron pairs to the central metal ion.
a. Four ammonia molecules donate four electron pairs to Cu2+ ion to form the complex cuprammonium ion, [Cu(NH3)4]2+.
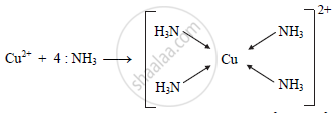
b. Six CN- ligands donate six electron pairs to Fe2+ and Fe3+ ion to form the complex ion ferrocyanide, [Fe(CN)6]4- and ferricyanide, [Fe(CN)6
APPEARS IN
RELATED QUESTIONS
Illustrate with example, the difference between a double salt and a co-ordination compound
Write ‘four’ characteristics of co-ordinate complex ion.
How ligands are classified? Explain with suitable examples.
What is effective atomic number? Calculate effective atomic number of copper ( Z = 29 ) in (Cu ( NH3)4)2+
