Advertisements
Advertisements
Question
Explain the following:
Electrovalent compounds conduct electricity.
Short Note
Solution
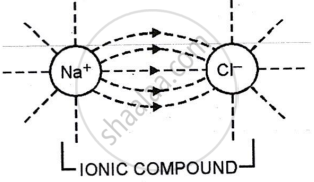
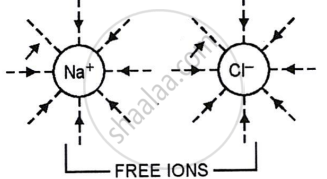
Electrovalent compounds in aqueous or fused states conduct electricity due to the breaking of electrovalent bonds between oppositely charged ions. Ions can move freely in all directions. When an electric potential is supplied to a solution, cations migrate to the cathode and anions to the anode, resulting in an electric current.
shaalaa.com
Is there an error in this question or solution?
